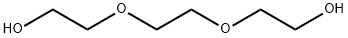| Chemical Properties |
Triethylene glycol is a clear, colorless, viscous, stable liquid with a slightly sweetish odor. Soluble in water; immiscible with benzene, toluene, and gasoline. Combustible. Because it has two ether and two hydroxyl groups its chemical properties are closety related to ethers and primary alcohols. It is a good solvent for gums, resins, nitrocellulose, steam-set printing inks and wood stains. With a low vapor pressure and a high boiling point, its uses and properties are similar to those of ethylene glycol and diethylene glycol. Because it is an efficient hygroscopic agent it serves as a liquid desiccant for removing water from natural gas. It is also used in air conditioning systems designed to dehumidify air. |
| Uses |
triethylene glycol is a solvent prepared from ethylene oxide and ethylene glycol.
Triethylene glycol can be used:
To prepare fatty acid gelators, which are used to gelate various edible and vegetable oils.
As a solvent to prepare superparamagnetic iron oxide nanoparticles for in situ protein purification.
As an absorbent agent in the subsea natural gas dehydration process. |
| Uses |
In various plastics to increase pliability; in air disinfection. |
| Uses |
Triethylene glycol is used as a plasticizer, as an additive for hydraulic fluids and brake fluids, and as a disinfectant. It is an active component of certain pigments, printing dyes, inks and paste. It finds application as a liquid desiccant and used in the dehydration of natural gas, carbon dioxide, hydrogen sulfide and air conditioning systems. It plays as an important role in anti-freeze and de-icing products, cleaning and furnishing care products, lubricant and greases. |
| Definition |
ChEBI: Triethylene glycol is a poly(ethylene glycol) that is octane-1,8-diol in which the carbon atoms at positions 3 and 6 have been replaced by oxygen atoms. It has a role as a plasticiser. It is a poly(ethylene glycol), a diol and a primary alcohol. |
| Production Methods |
Triethylene glycol, like diethylene glycol, is produced commercially as a by-product of ethylene glycol production. Its formation is favored by a high ethylene oxide to water ratio. |
| General Description |
Triethylene Glycol (TEG) is a colorless liquid with a mild odor and is denser than water. It is a member of a series of dihydroxyalcohols. TEG's primary applications are derived from its ability to absorb moisture. It is utilized as a dehydrating agent in natural gas pipelines to eliminate water from the gas before reusing it in the system. Triethylene glycol also has mild disinfectant qualities and, when volatised, is used as an air disinfectant for virus and bacteria control. |
| Reactivity Profile |
Triethylene glycol is a ether-alcohol derivative. The ether being relatively unreactive. Flammable and/or toxic gases are generated by the combination of alcohols with alkali metals, nitrides, and strong reducing agents. They react with oxoacids and carboxylic acids to form esters plus water. Oxidizing agents convert alcohols to aldehydes or ketones. Alcohols exhibit both weak acid and weak base behavior. They may initiate the polymerization of isocyanates and epoxides. Reacts with strong oxidants. [Handling Chemicals Safely 1980. p. 932]. |
| Health Hazard |
Under normal conditions of use, Triethylene Glycol (TEG) is not expected to cause irritation to the skin, eyes or respiratory tract. However, in applications where vapours or mists are created, inhalation may cause irritation to the respiratory system.
No ceiling on worker exposure has been set by the American Conference of Governmental Hygienists (ACGIH), neither has a Workplace Exposure Limit been established for TEG. TEG is readily biodegradable, has a low potential to bioaccumulate and has low toxicity to aquatic organisms.
TEG is not flammable, unless preheated. |
| Flammability and Explosibility |
Not classified |
| Safety Profile |
Poison by intravenous route. Mildly toxic to humans by ingestion. Experimental reproductive effects. An eye and skin irritant. Many glycol ether compounds have dangerous human reproductive effects. Combustible when exposed to heat or flame. Can react with oxidizing materials. Explosive in the form of vapor when exposed to heat, flame, or spark. To fight fire, use alcohol foam, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also ESTERS and GLYCOL ETHERS. |
| Purification Methods |
Dry the glycol with CaSO4 for 1 week, then it is repeatedly and very slowly fractionally distilled under a vacuum. Store it in a vacuum desiccator over P2O5. It is very hygroscopic. [Beilstein 1 IV 2400.] |