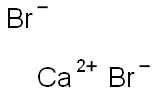| Uses |
Calcium bromide (CaBr2) is used as a developer for photographic film and paper and as a dehydrating agent (drying agent), food preservative, and fire retardant. |
| Description |
Calcium bromide has the chemical formula CaBr2. It was first prepared by Pfeiffer, Klossmann and Angern in 1924. It is a deliquescent salt that has the form of colorless hexagonal crystals that are soluble in water and absolute alcohol. Its molecular weight is 199.90 g/ mol for the anhydride. When strongly heated in air, calcium bromide will produce CaO (lime) and Br2. It is generally obtained as the dihydrate, CaBr2·2H2O, in the form of a white crystalline powder. A hexahydrate, CaBr2·6H2O, is also known. Anhydrous calcium bromide is obtained by dehydration (250°C, vacuum) of the dihydrate CaBr2·2H2O. On contact with hot surfaces or flames, CaBr2 decomposes forming bromine as a toxic and corrosive fume. It also reacts with strong acids to produce corrosive fumes of bromine.
In its solid anhydrous state, it is a white powder. CaBr2 can be found in drilling muds, neuroses medication, freezing mixtures, food preservatives, photography and fire retardants. The anhydride has a melting point of 730°C and a boiling point of 810°C where it decomposes. Its density is 3.41 g/cm3. It is moderately soluble in water at 142 g/100 ml of water. |
| Chemical Properties |
Calciumbromide, CaBr2, is a colorless crystalline solid with a melting point of 765°C. It is deliquescent and is soluble in water and absolute alcohol. Calcium bromide is used in medicine. Calcium bromide(hydrated), CaBr2·3H20, has a meiting point of 80.5℃. |
| Uses |
In photography for making dry plates and light-sensitive papers; manufacture of mineral waters, NH4Br, fire-extinguishing compositions. |
| Uses |
Made by the action of hydrobromic acid on calcium oxide and crystallization. The white granular crystals are soluble in water. Calcium bromide was used in making collodion emulsions, particularly for dye-sensitized plates. |
| Uses |
Calcium bromide has a number of useful applications. Used in the oil and gas fields, the compound is a true working example. At almost every stage of the oil and gas extraction process, calcium bromide has found extensive usage. Stability, dependability and ease of use distinguish calcium bromide in the drilling process. There, it is used in "drilling muds" worldwide. |
| Preparation |
CaBr2·2H2O has a melting point of 38°C where it dissolves in its own waters of hydration. Its density is 2.290 g/cm3. It can be prepared by reaction in solution with HBr on the carbonate:
CaCO3 (solid)+HBr (liq)→CaBr2 (liq)+ CO2 (gas)
An improved method for producing calcium bromide has been developed by reacting hydrogen bromide with calcium hydroxide in the presence of water. |
| Definition |
ChEBI: Calcium dibromide is a calcium salt. |