Product Introduction
| Cresyl glycidyl ether Basic information |
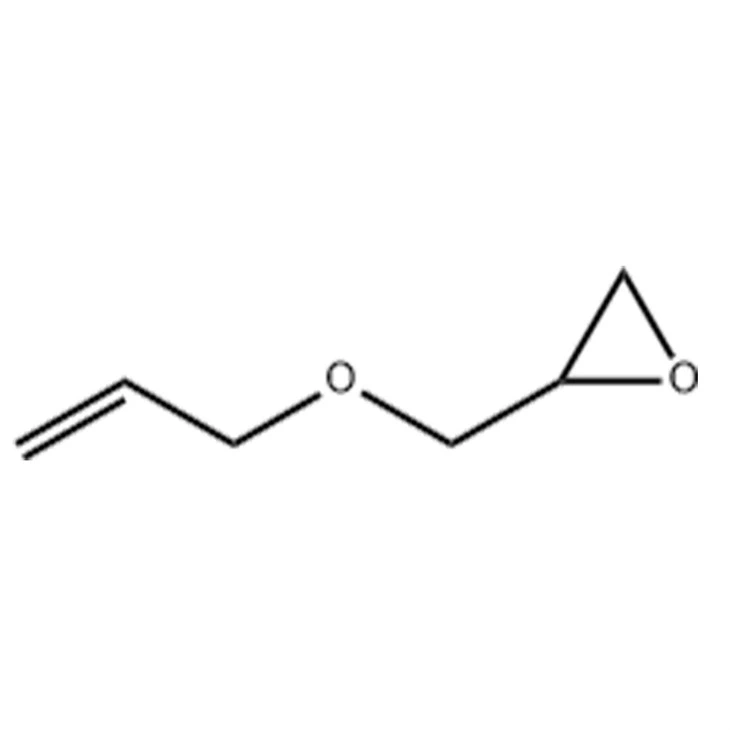
| Product Name: | Cresyl glycidyl ether |
| Synonyms: | ((methylphenoxy)methyl)-oxiran;[(methylphenoxy)methyl]-oxiran;1,2-epoxy-3-(tolyloxy)-propan;2,3-epoxypropyltolylether;cresolglycidalether;cresolglycidylether;cresylglycideether;cresylglycidylether(mixedisomers) |
| CAS: | 26447-14-3 |
| MF: | C10H12O2 |
| MW: | 164.2 |
| EINECS: | 247-711-4 |
| Mol File: | 26447-14-3.mol |
| Cresyl glycidyl ether Chemical Properties |
| density | 1.14 |
| Water Solubility | <=0.1 g/100 mL at 21 ºC |
| Stability: | Stable. Incompatible with strong acids, bases and oxidizing agents. May attack some types of plastic. Combustible. |
| EPA Substance Registry System | Cresyl glycidyl ether (26447-14-3) |
| Safety Information |
| Hazard Codes | Xn |
| Risk Statements | 61-36/38-43-51/53-62 |
| Safety Statements | 53-26-36/37-45-61 |
| RIDADR | UN 3082 9/PG 3 |
| WGK Germany | 3 |
| F | 21 |
| Hazardous Substances Data | 26447-14-3(Hazardous Substances Data) |
| Provider | Language |
|---|---|
| 1,2-Epoxy-3-(tolyloxy)propane | English |
| Cresyl glycidyl ether Usage And Synthesis |
| Description | Cresyl glycidyl ether is a reactive diluent added in epoxy res ins of the Bisphenol A type. |
| Chemical Properties | colourless liquid |
| General Description | Colorless liquid. Sinks and mixes with water. |
| Air & Water Reactions | Oxidizes readily in air to form unstable peroxides that may explode spontaneously [Bretherick, 1979 p.151-154, 164]. Insoluble in water. |
| Reactivity Profile | A phenol and epoxide. Phenols do not behave as organic alcohols, as one might guess from the presence of a hydroxyl (-OH) group in their structure. Instead, they react as weak organic acids. Phenols and cresols are much weaker as acids than common carboxylic acids (phenol has Ka = 1.3 x 10^[-10]). These materials are incompatible with strong reducing substances such as hydrides, nitrides, alkali metals, and sulfides. Flammable gas (H2) is often generated, and the heat of the reaction may ignite the gas. Heat is also generated by the acid-base reaction between phenols and bases. Such heating may initiate polymerization of the organic compound. Phenols are sulfonated very readily (for example, by concentrated sulfuric acid at room temperature). The reactions generate heat. Phenols are also nitrated very rapidly, even by dilute nitric acid. Epoxides are highly reactive. They polymerize in the presence of catalysts or when heated. These polymerization reactions can be violent. Compounds in this group react with acids, bases, and oxidizing and reducing agents. They react, possibly violently with water in the presence of acid and other catalysts. |
| Health Hazard | Contact with eyes causes irritation. Contact with skin causes primary irritation and allergic sensitization. |
| Fire Hazard | Special Hazards of Combustion Products: Wear full body and respiratory protection. |
| Contact allergens | It is a reactive diluent added in epoxy resins Bisphenol A type. |
Hot Tags: cresyl glycidyl ether, China cresyl glycidyl ether manufacturers, suppliers, factory, Octadecyl Acrylate, CAS 5888 33 5, CAS 4813 57 4, CAS 66492 51 1, Stearyl Methacrylate, CAS 109 16 0
Previous: Allyl Glycidyl Ether
Next: Butyl Glycidyl Ether
You Might Also Like
-

Electronic Grade 99.9% Purity Diethylene glycol dimethyl Ether/DEDM CAS 111-96-6
-

Factory Supply Ethylene Glycol Di-Acetate/EGDA CAS 111-55-7
-

Colorless Liquid 99% Dipropylene Glycol Monomethyl Ether Acetate/DPMA CAS 88917-22-0
-

CAS 34590-94-8 Dipropylene glycol monomethyl ether/DPM
-

Hmp 536ba 531ba Polyoxypropylene Bisphenol-a Ether CAS 32492-61-8
-

Factory Supply Poly (ethyl vinyl ether) CAS 25104-37-4
Send Inquiry

