| Description |
The cyanopyridines are as follows:2-cyano-: A white to tan liquid or solid with an almondodor. Boiling point=about 213℃; Freezing/Meltingpoint=27℃; Flash point=89℃.3-cyano-: A colorless liquid or gray crystalline solid.Molecular weight=104.12; Boiling point=83-84℃;Freezing/Melting point=47-49℃. Hazard Identification(based on NFPA-704 M Rating System): Health 3,Flammability 3, Reactivity 0. Soluble in water.4-cyano-: A beige solid. Freezing/Melting point=75.8℃ |
| Chemical Properties |
white to brown low melting crystalline solid |
| Chemical Properties |
The cyanopyridines are as follows: 2-cyano-: A white to tan liquid or solid. Almond odor. Boiling point=2213℃ ; freezing/melting point=27℃ ; flash point=89℃ . 3-cyano-: a colorless liquid or gray crystal- line solid. |
| Uses |
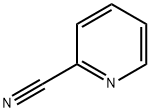
2-Cyanopyridine is a cyano substituted pyridine. 2-Cyanopyridine is a related compound of nicotine and is a component of tobacco smoke condensate. |
| Uses |
It is employed as an important chemical intermediate for rimiterol hydrobromide and used as a bronchodilator. It is also used as intermediates of pharmaceutical and dye and pigment. It acts as a precursor of the respective amidate for protein modification via amidation. |
| Definition |
ChEBI: A cyanopyridine carrying the cyano group at position 2. |
| Potential Exposure |
Limits in Air NIOSH IDLH525 mg/m3 NIOSH REL: (nitriles) 2 ppm, Ceiling Concentration, not to be exceeded in any 15-minute work period. |
| First aid |
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit. |
| storage |
Color Code-Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with thischemical you should be trained on its proper handling andstorage. Store in tightly closed containers in a cool, wellventilated area away from heat or flame and separate fromoxidizing materials |
| Shipping |
UN3276 Nitriles, liquid, toxic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required, Potential Inhalation Hazard (Special Provision 5). |
| Incompatibilities |
Oxidizing agents, such as perchlorates, peroxides, and permanganates. Nitriles may polymerize in the presence of metals and some metal compounds. They are incompatible with acids; mixing nitriles with strong oxidizing acids can lead to extremely violent reactions. Nitriles are generally incompatible with other oxidizing agents such as peroxides and epoxides. The combination of bases and nitriles can produce hydrogen cyanide. Nitriles are hydrolyzed in both aqueous acid and base to give car- boxylic acids (or salts of carboxylic acids). These reactions generate heat. Peroxides convert nitriles to amides. Nitriles can react vigorously with reducing agents. Acetonitrile and propionitrile are soluble in water, but nitriles higher than propionitrile have low aqueous solubility. They are also insoluble in aqueous acids . |