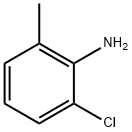
| Description |
2-chloro-6-methylaniline is an important organic synthesis intermediate widely applied to chemical pharmacy, pesticides, and organic synthesis. In the aspects of chemical pharmacy and pesticides, 2-chloro-6-methylaniline can be used as a reaction starting material to participate in the preparation processes of tyrosine kinase inhibitors dasatinib, indazole derivatives with anti-breast cancer activity, 2-methyl-6-chlorophenyl sulfonylurea derivatives with bacteriostatic activity and the like; in the aspect of organic synthesis, 2-chloro-6-methylaniline can introduce chemical groups such as hydrazine, nitro, cyano, halogen (F, Cl, Br), and the like into an aromatic ring through diazotization reaction, thereby participating in wide chemical reactions. |
| Chemical Properties |
clear yellow to red-brown liquid |
| Chemical Properties |
The chloromethylanilines are colorless or white crystalline solids or liquids, some have a mild fishy odor. |
| Uses |
2-Chloro-6-methylaniline is a substituted aniline used in the preparation of various pharmaceutical compounds. 2-Chloro-6-methylaniline showed inhibition of the polymerization of vinyl acetate. |
| Preparation |
A preparation method of 2-chloro-6-methylaniline takes 3-chloro-5-methyl-4-nitroaniline as a starting material, water as a solvent, and sulfuric acid as a reactant. Eliminates amino groups through diazotization reaction, reduces by hypophosphorous acid to obtain an intermediate, and finally takes iron powder as reducing nitro groups to prepare the 2-chloro-6-methylaniline through a one-pot reaction. |
| Potential Exposure |
Most of the isomers are used in dyestuff manufacture. The 3-chloro-para isomer is used to kill birds. It is marketed as pelleted bait for control of bird populations. |
| Shipping |
UN2239 Chlorotoluidines, solid, Hazard Class: 6.1; Labels: 6.1-Poisonous materials. UN3429 Chlorotoluidines, liquid, Hazard Class: 6.1; Labels: 6.1- Poisonous materials |
| Incompatibilities |
Incompatible with oxidizers, strong acids; chloroformates, and acid anhydrides, isocyanates, aldehydes forming fire and explosive hazards. |