| Chemical Properties |
clear colourless to light yellow liquid |
| Uses |
Diallyl Phthalate is used as a reagent in ring-closing ruthenium based reactions. |
| Preparation |
Diallyl phthalate (DAP) is prepared by reaction of phthalic anhydride and allyl alcohol:
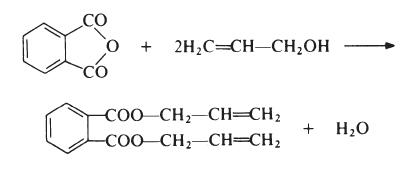
|
| Application |
Diallyl phthalate is an important monomer for the production of thermosetting molding compounds, which must have good dimensional stability and electrical properties, and be resistant to heat and solvents. Diallyl phthalate can be polymerized or copolymerized. This usually is done by dissolving the diallyl phthalate monomer in 2- propanol, adding 50% hydrogen peroxide at about 105 ℃, and precipitating the prepolymer from the cooled, viscous solution with excess 2- propanol. Copolymers containing diallyl phthalate are suitable for specialty coating and for embedding, especially in the production of electronic devices. For example, the moisture-sensitive epoxy compounds now used in light-emitting diode (LED) displays can be replaced by stable diallyl phthalate epoxy encapsulating resins. By adding inorganic materials to diallyl phthalate prepolymer compositions, reinforced thermosetting molding compounds can be obtained. Glass cloth or paper can be impregnated with a solution of prepolymer, monomer, and peroxide initiator. After removal of the solvent, the glass cloth or paper is cured to give the desired film-protected material, which is used for decoration, stain-resistant overlays for household articles, and furniture. |
| General Description |
Clear pale-yellow liquid. Odorless. |
| Air & Water Reactions |
Incompatible with water and oxygen. Should be stored air tight, with inhibitor, to prevent polymerization reaction . |
| Reactivity Profile |
Diallyl phthalate can react with oxidizers. Diallyl phthalate can also react with acids and alkalis. Diallyl phthalate is incompatible with water and oxygen. |
| Fire Hazard |
Diallyl phthalate is combustible. |
| Flammability and Explosibility |
Not classified |
| Safety Profile |
Suspected carcinogen with experimental carcinogenic data. Moderately toxic by ingestion, skin contact, intraperitoneal, and subcutaneous routes. An eye irritant. Mutation data reported. Combustible when exposed to heat or flame; can react with oxidzing materials. To fight fire, use CO2 or dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also ALLYL COMPOUNDS and ESTERS. |
| Carcinogenicity |
In the 103-week study referred to previously, a slight increase in MNCL was seen in female rats treated with 50 or 100 mg/kg/day of DAP. MNCL occurs in F344 control rats at a high incidence; however, the incidence of 51% in female rats at the high dose level was above historical control data for the laboratory (29%). No significant increases in tumor incidences were seen in male rats. Based on this study, DAP was considered to have demonstrated equivocal evidence for carcinogenicity in female F344 rats according to the NTP.
In male and female B6C3F1 mice receiving 300 mg/kg of DAP by gavage for 103 weeks (5 days/week), the incidence of forestomach papillomas was significantly greater than that of controls. Because of the rarity of forestomach papillomas in control B6C3F1 mice and the concomitant observation of dose-related forestomach hyperplasia, the development of these tumors was considered to be test substance related. Compared to controls, a slight increase in the incidence of lymphomas was observed in males receiving 300 mg/kg/day of DAP. Because the increase was not statistically significant compared to historical control data, this effect was not considered to be test substance related. |








![2-Propenoic Acid, 2-[2-(ethenyloxy)ethoxy]ethyl Ester](/uploads/41226/small/2-propenoic-acid-2-2-ethenyloxy-ethoxy-ethyl3d579.jpg?size=336x0)
